BayMedica LLC: Working With Cannabis Multi-State Operators To Make Their Products Better
InMed Pharmaceuticals Inc. (Nasdaq: INM), a leader in the research, development, manufacturing and commercialization of rare cannabinoids, is taking a leading position in the marketplace. Less that a month after launching B2B sales of the emerging rare cannabinoid delta 9-dominant tetrahydrocannabivarin (d9-THCV) into the health and wellness sector, we interviewed BayMedica LLC Vice President of Sales, Jerry Griffin, how the business is going. BayMedica is the wholly-owned subsidiary of InMed Pharmaceuticals, acquired last October.
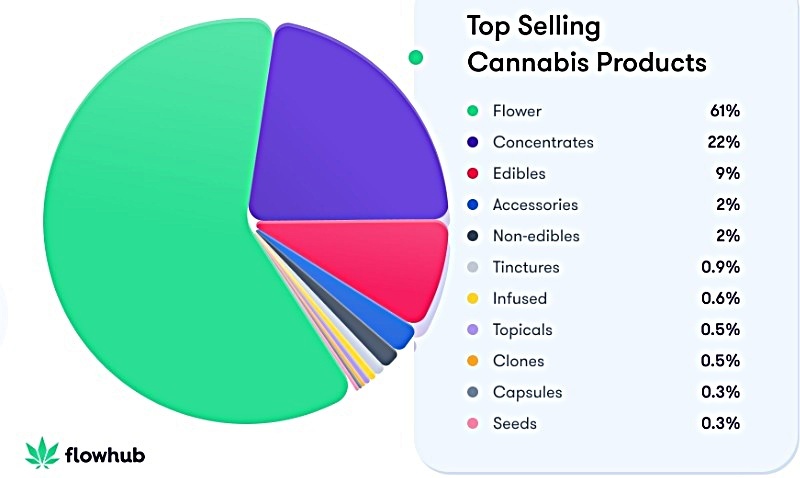
While BayMedica is conducting business activities with many different types of health and wellness companies, Jerry gives an interesting account about the interest coming from U.S. multi-state operators (MSOs) in the cannabis industry. To help optimize the effectiveness of their products—to counterbalance the effects of THC, for example—MSOs are increasingly infusing cannabinoids into edibles formulations. Edibles currently account for about 9% of the U.S. cannabis market, and that metric has been steadily rising over time.
As Jerry Griffin explains: “So we talk to a lot of MSOs. We talk to a lot of single-state operators. A lot of them ae looking at rare cannabinoids simply to make their existing products better.”
For definition purposes, a multi-state operator can be defined as a company that sells cannabis products in a given state that must also be grown, processed and packaged in that jurisdiction. Multi-state operators typically set up multiple subsidiaries that operate throughout the United States where cannabis is legal, whereas single-state operators are generally smaller enterprises which operate in one particular state.
The infusion of rare cannabinoids into edible products is one of many avenues which is expected to stoke growth for the next several years. According to a recent research study, the U.S. minor cannabinoids market size & share is expected to grow at a CAGR of 23.4% between 2022 and 2030. The U.S. minor cannabinoids industry revenue of USD 5.1billion in 2021 is expected to grow up to USD 28.9 billion by 2030.
Research firm Edison Group has previously predicted that the global market for CPG and pharma cannabinoid biosynthesis products could reach $92 billion by 2040.
Not just limited to the ingestion of pills or edibles, cannabinoids can be applied topically as well. Last year, InMed Pharmaceuticals initiated a clinical trial for analyzing the utilization of CBN-based topical cream for the treatment of Epidermolysis Bullosa. The disease is the collective name of a group of genetic disorders of characterized by fragile skin and mucous membranes that are easily damaged, leading to extensive blistering and wounding. The study, currently in Phase 2, remains active.
Click on the embedded link to see the full interview with BayMedica LLC Vice President of Sales, Jerry Griffin, in his own words.



